SCOMNET ( CoVid Medical Device Producer ) + Ace Market Melting Up
InsiderShark
Publish date: Fri, 07 Aug 2020, 04:48 PM
Join our Telegram Channel BursaBets
Supercomnet [ 0001 ] said its latest catheter for cardiovascular use has been approved and is now listed on the US Food and Drug Administration's (FDA) website. The FDA approval means that the company can now start manufacturing the catheter for its Denmark-based client Ambu and also New York Stock Exchange-listed Edwards Lifesciences Corp, both of which have signed long-term agreements with the company. Orders increased by 5% to 10% every year, whether or not there was a recession.
The main products developed by the company include disposable medical consumables for Class 1, Class 2 and intensive care unit (ICU) usage. They are entering a new market, means they are growing and expanding. Revenue will go up, this means higher stock price in the coming future. World population continues to grow, we are entering an aging society, this means more medical needs.
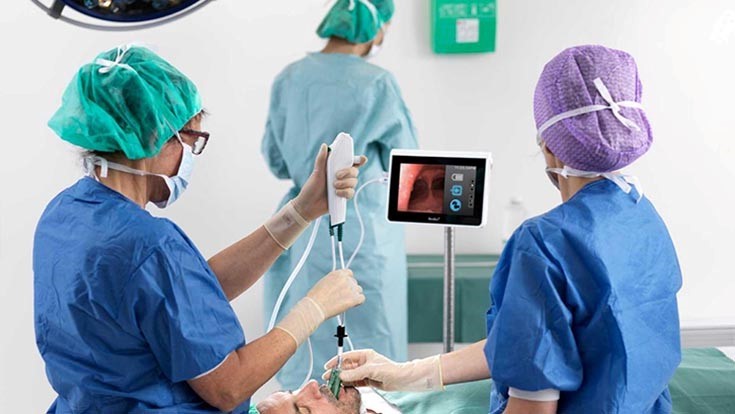
"It is worth noting that several key products of SMP are utilised in critical medical devices that are needed in the treatment of Covid-19 patients. Among the critical medical devices are single use disposable bronchoscopes, which are used in the first line of lung treatment for patients with suspected or confirmed Covid-19 infections, and the critical care monitoring cable that is presently being used for patients in the intensive care unit." Source: NST
Scomnet is a multiyear healthcare growth stock. It’s attractive also because there are not many heathcare stocks in the region that produces unique high-value medical products. Growth will be driven by its two main customers: US-listed Edwards Lifesciences Corp and Denmark-listed Ambu. To put in perspective, Edward Lifesciences is a company with a market cap of US$42 bil.
-
Supercomnet Technologies wired to medical device market growth
-
Supercomnet hits record high as orders from medical device makers soar
- Why We’re Not Concerned About Supercomnet Technologies Berhad’s Share Price

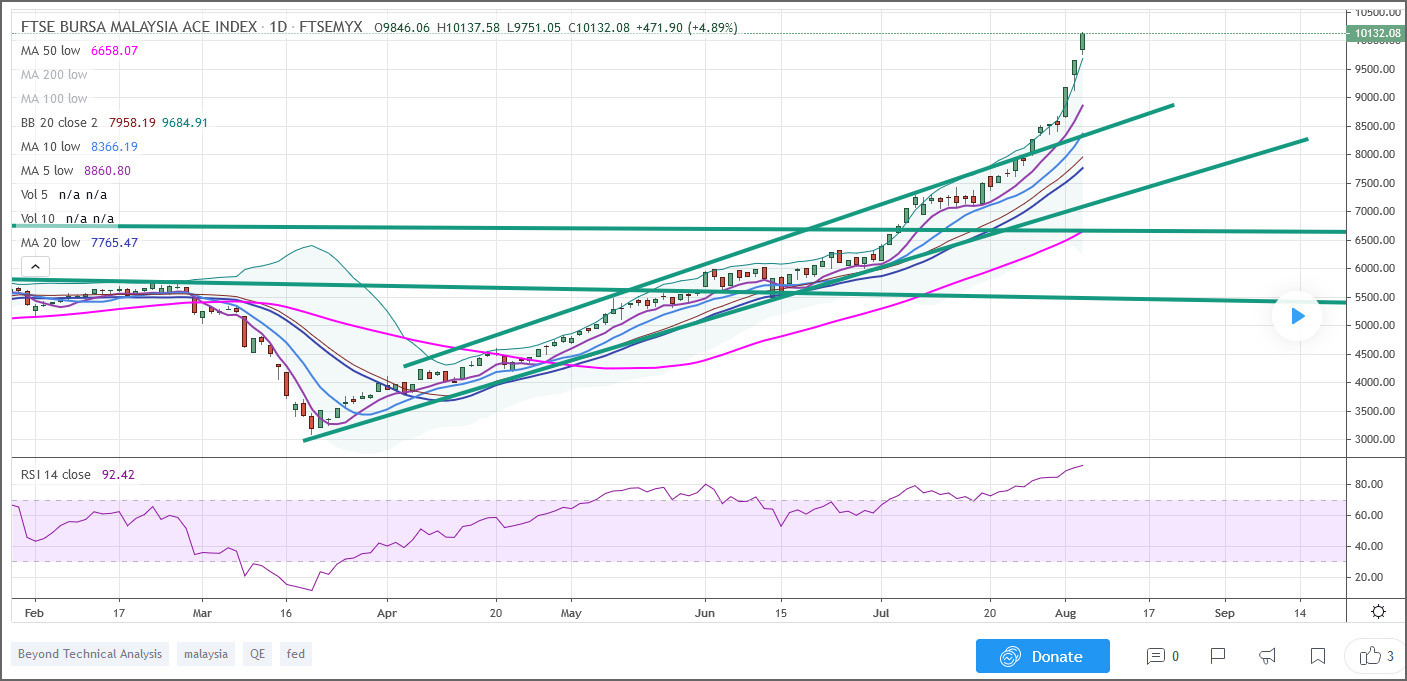
Will Supercomnet price collapse if the bubble burst? Yes, it will go down together with the rest, that is how it is. For the moment, the rally appears intack and the upside is bigger than the downside. Trade or invest safely, remember, always do your own research.
Ace Market Melting Up - Ride The Bull
More articles on BursaBets [ Rabbit Trader Turtle Investor ]
Created by InsiderShark | Oct 01, 2020
Created by InsiderShark | Jul 20, 2020





















